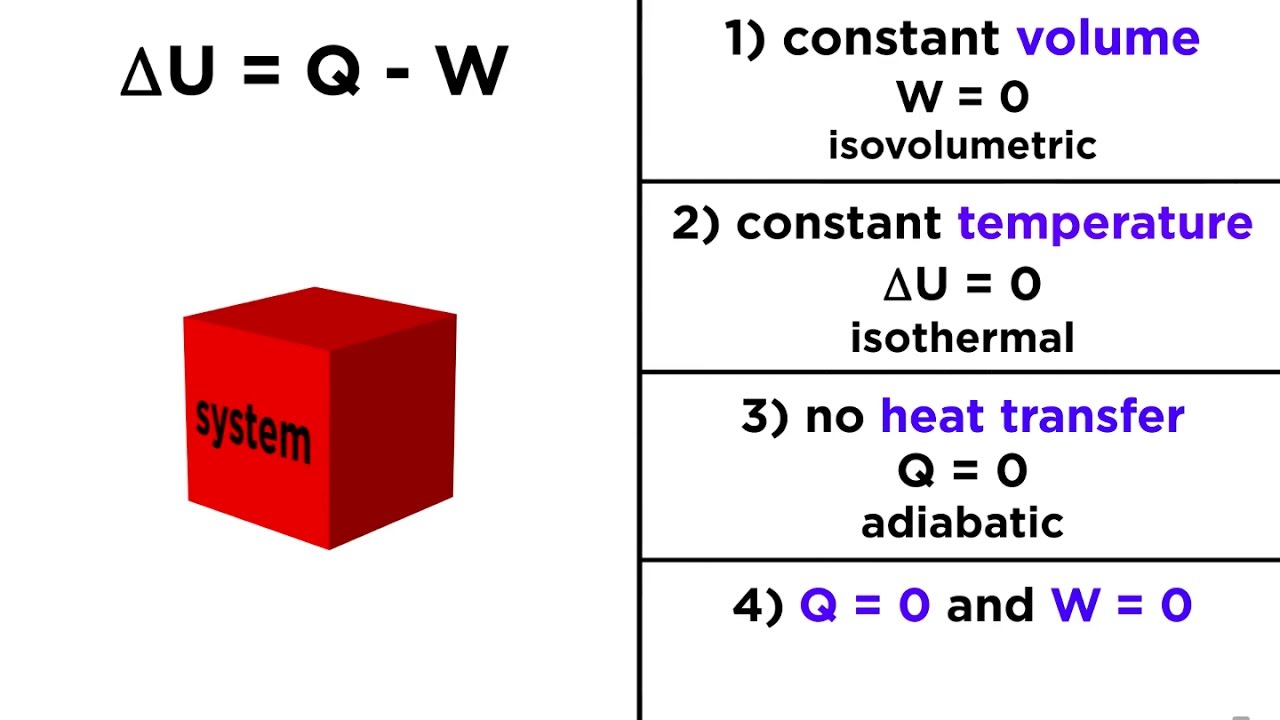
In the same way, the energetic state of a gas at a temperature T can be changed by transferring energy – it can be brought to a higher temperature. The energetic state of the object located at a height h can be changed by transferring energy – it can be brought to a higher height. In the case of internal energy, the relationship between internal energy U and temperature T is established by the specific isochoric heat capacity c v (dependent on the type of gas!). 
The exact relationship between the potential energy and the height h is established by the gravitational acceleration g (dependent on the position!). In the same way, a certain internal energy can be assigned to a given temperature T via the mass m of the gas. Figure: Analogy between potential energy and internal energyĪ given height h can be assigned a certain potential energy via the mass m of the object. While the gravitational potential energy represents the energetic (gravitational) state of an object at a given height h, the internal energy represents the energetic (kinetic) state of an ideal gas at a given temperature T. Internal energy of ideal gases can very clearly be compared in analogy to gravitational potential energy of an object. First of all, select the option either you want to find heat energy, specific heat, mass, initial temperature, final temperature of any substance Next, select. The three variables are related by the equation. It says: The increase in internal energy of a thermodynamic system is equal to the heat added to the system plus the work done on the system. The heat capacity, C, is the amount of heat, q, required to raise the temperature, T, of an object by 1 o C. This statement forms the base of the First Law of Thermodynamics. Analogy between internal energy and gravitational potential energy The First Law of Thermodynamics We stated earlier that the two ways to transfer energy to or from a system are doing work and exchanging heat. &\boxed) still apply without restriction. According to the first law of thermodynamics, this internal energy can be changed by transferring energy as work W or as heat Q: In the article Internal e nergy of ideal gases it was explained in detail that in ideal gases only the kinetic energy of the gas molecules exists as internal energy (thermal energy).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
